Let’s talk about sex.
Hot and heavy sex, for cold-blooded fish at least. The kind where a female commands a male to do her bidding, locks him into weeks of servitude, casts him aside like yesterday’s flotsam and moves on to the next guy. Every year in the northwest Pacific, this happens to male after helpless male at the hands (fins?) of a species of all-female fish—the piscines of Themyscira, so to speak.
It is one of the most fascinating and thought-provoking forms of reproduction I’ve ever come across. I’m itching to tell you about it.
But first, let me introduce you to Kevin.
It was October 2019. The skies were clear. Winds and water calm, the ocean somewhat green, as is often the case in northern seas. The temperature underwater was about 8ºC (about 46ºF), which was actually much warmer than I had anticipated.
The last time I had been standing in this very spot, the sea had been 3ºC (about 37ºF), so the ocean on this day seemed almost tropical. OK, not really, but it wasn’t too bad. Given that I was about to submerge myself in cold water yet again, the prospect of somewhat cold was better than #^@$*!!! cold.
Being relatively new to diving with a drysuit, I was slow getting myself together. There is so much stuff.
After years of needing not much more than a 1mm shorty and my mask+snorkel+fins, the process of donning insulating layers, zipping up my drysuit, checking seals, squishing my head into a hood, clipping on ankle weights (note to self—something which should really be done prior to putting on thick gloves that make for fat and fumbly fingers), a vest full of lead on my chest and more on around my waist, followed by checking of dive computer, turning on of fiddly camera bits, and finally, walking into the water to put on fins while not losing photo and lighting paraphernalia—well, let’s just say it took a while. Still does.
My friend and guide Sato-san was already in the water, waiting for the slowpoke newbie. He is a veteran, having dived in these conditions nearly every day for the better part of three decades. Thus were my onshore antics of no interest, not even mild entertainment value.
Our objective that day was to visit this fish:
I’ll wager that you’ve never seen this animal, unless you live in Japan, Korea, China or some remote parts of southeast Russia.
It is a fat greenling, known as アイナメ (pronounced ai-na-meh) in Japanese, called アブラコ (pronounced ah-bu-rah-ko) in the local dialect of Hokkaido, and vested with the binomial moniker of Hexagrammos otakii.
Mature adults can reach something around 60cm in length (almost 24 inches), weigh up to 2.4kg (5.3lbs) or so. They generally frequent deeper water along rocky, coastal areas, down to 150m-ish (492ft-ish).
If you Google the name and look at photos, you’ll see that these fish are usually tan, brown, and sand-coloured, mottled in pattern, drab and…well…meh in overall appearance.
So why—you might ask—is this particular fish such a brilliant yellow-orange?
Because he’s horny.
Each season, at different times depending on the location, members of this species ascend to shallow water for reproduction. Males like this one construct and maintain nesting sites, a process that entails preparing a hard surface, such as a rock adorned with nice grass and/ or algae patches, to which they attempt to entice females using a combination of colour (hence the chic yellow), seduction and sometimes mild coercion.
Some males are really good at this; while others sort of suck at it.
Popular males find themselves the center of female attention, evidenced by multiple clutches of eggs. Less successful ones may find themselves with few or no female takers and/ or pushed to the outer edges of nesting areas. They may also end up losing what eggs they have to interlopers (i.e., other fish) that raid nests for tasty, nutritious snacks.
Look closely at the photo above, and you’ll see that this dapper fellow has been successful in his efforts. He is atop a sizeable cluster of eggs that are almost ready to hatch. Below is a photo of the fish tending to his precious eggs.
I didn’t know it at the time, but this was my first encounter with Kevin, a fish with whom I would later develop a wonderful, enlightening friendship.
In normal sexual reproduction, genetic material from a male combines with that from a female to form a new individual. In science-speak, two haploid cells called gametes (sperm and egg) fuse to make a diploid cell (zygote). That’s how you and I originated. That’s how most animals, including fish, come into being.
Nature being wild and crazy though, there are exceptions and variations to this recipe.
Ants, bees and wasps for example, exhibit what’s called haplodiploidy (Try saying that five times in rapid succession!), which translated to normal-speak means that females have a complete set of genetic material, while males are half-baked (you smirked, didn’t you?).
In general though, one would bet on the fully formed and functional male/ female scenario when exchange of genetic material is involved, each generation passing on genes inherited from both parents to the next generation.
Then there is asexual reproduction, which means making babies without the need for genetic recombination. In a way, this is more efficient, as there is no need to hang out at bars to find a willing partner (oops, perhaps I’ve said too much). But the potential downside is lower genetic variation when compared to sexual reproduction. That could manifest as diminished resilience on a population/ species level when faced with problem situations, like say, a pandemic.
Take parthogenesis as an example. This term describes the scenario in which an embryo develops from an unfertilised egg—meaning no males involved whatsoever. Nada. This happens with some plants, some invertebrates like nematodes and aphids, and even occasionally in some vertebrates—including reports of fish, amphibian, reptile and bird species.
Gynogenesis is similar, but this type of asexual reproduction needs an extra step. Sperm from a male is required to kickstart the process. But the male’s DNA does not combine with the female’s, so there is no genetic contribution from the male.
Which brings us to Kevin. Or not to Kevin specifically, but to his genus.
As mentioned above, the binomial name for fat greenlings is Hexagrammos otakii, with Hexagrammos denoting the genus. As a brief reminder from high school biology class (I sense some cringing going on), this name derives from the Linnean system of taxonomy, i.e., the naming and cataloging of life on Earth.
One of the primary reasons we use this naming convention (which can seem like gibberish if you’re not familiar with it) is that the Linnaean framework makes it possible for everyone around the world to know exactly which organism is being discussed.
Instead of asking, “Hey, is a fat greenling and an aburako the same thing?” it is possible just to observe: “That is Hexagrammos otakii,” and there will be no ambiguity.
There is an additional feature that derives from this system.
The basic structure of this taxonomic hierarchy goes: Kingdom, Phylum, Class, Order, Family, Genus, Species (with more sub-categories within each major category). By convention, all terms except the species name are capitalised and are often printed in italics.
For Kevin and his kin then, the Linnean nomenclature would be:
Kingdom: Animalia
Phylum: Chordata
Class: Actinopterygii
Order: Scorpaeniformes
Family: Hexagrammidae
Genus: Hexagrammos
Species: otakii
Think of this as being sort of like a family tree. The further up the chain you go to find commonality, the more distant the relation. Conversely, the lower down the chain, the closer you are.
This is where things get interesting.
Fast-forward to almost exactly a year later, and there I am again, fumbling with zippers and gear, preparing to immerse myself in cold water. I am back in Hokkaido, the northernmost of Japan’s four main islands, to search for fat greenlings. It is spawning season.
The sea is slightly warmer than it was the last time I had visited. In many places around Japan, the temperature of the oceans has been higher than normal the entire year. Every place I’ve been, the story has been the same: “Things are different; the animals are late,” and so forth.
Fat greenlings were no exception. There weren’t many around yet. The reproductive season was off to a slow start.
I made my way across sand and rubble to where there were large rocks and seagrass—favoured topography for otakii nests.
Sato-san had already scouted the area. We went directly to a greenling. The bright yellow of the male popped like neon against the blue of the sea.
He had chosen his rock well. It was big. There was a healthy patch of seagrass. The location afforded him an unimpeded 360-degree view. And he had cleaned the rock to perfection. How could a discerning female possibly resist?
One, possibly two, had already approved of his nest-keeping skills by depositing eggs. The large yellow fish lunged toward us with impressive speed, I assume as the beginning of a threat gesture. He diverted as soon as he recognised us.
It was Kevin!
Just to be clear, he wasn’t named Kevin yet. I gave him that name later, once we had cemented our relationship.
At that point, there was just something familiar about him. The way he moved. The way he patrolled. The way he gave us a once over, then accepted us.
In short—his personality.
By way of contrast, the two other males in the area were completely different. They had both picked what seemed to be less desirable nesting sites—cluttered, not as elevated, views partially obstructed (and hence more difficult to keep an eye out for potential egg-snatchers). One was attentive to his nest, but ducked for cover as soon as we approached. The third was even more of a scaredy-cat(fish). He disappeared completely, leaving his turf unguarded.
At this point, you might be thinking, “Hang on a second. You’re talking about fish. Fish don’t look after their babies!”
While this is true for the most part, about one in four species engages in some form of care for its offspring.(1) Cardinalfish and jawfish males, for instance, brood eggs in their mouths, caring for them until the baby fish hatch. Seahorse males brood developing babies in specialised pouches until the young are ready to pop out and face the world.
Notice in these examples that it is the males who take on the responsibility for protecting young. This is not always the case. With ghost pipefishes (Solenostomidae) for instance, it’s the females that hold and nurture developing eggs. But examples of paternal care are plentiful, with greenlings fitting this pattern.
Three species of the Hexagrammos genus live in these waters.
1. Hexagrammos octogrammus (“Hoc”); masked greenling; スジアイナメ (su-ji-ai-na-meh)
2. Hexagrammos agrammus (“Hag”); spotty-bellied greenling; クジメ (ku-ji-meh)
3. Hexagrammos otakii (“Hot”); fat greenling; アイナメ (ai-na-meh)
Their populations are not limited to just around Hokkaido, but their ranges overlap here, so they co-exist. Moreover, their reproductive seasons coincide. The figure below illustrates the geographical distribution:
You’ve already met Kevin, a representative of the otakii species, which is the biggest of the three.
For reference, this is what a male agrammus looks like:
This species attains a maximum length of about 30cm (about 11.8 inches, or half the size of a Kevin). Here is the same male looking after his eggs:
And here is a close-up of fresh agrammus eggs:
There was an octogrammus male in the vicinity looking after eggs as well, but he was in the surge zone. Conditions were never calm enough to photograph that individual. Picture a darker fish, bigger than the agrammus above (reaching a maximum length of around 42cm (16.5 inches).
These are the three characters in the evolutionary drama that I am going to attempt to explain—Hoc, Hag and Hot. Underscore the word “attempt,” because the research is relatively recent, and the plot intricate. Most of my information is sourced from research papers, kindly provided to me by the lead researcher Munehara-san. Any errors or other stupidity is due to me, not the researchers.
Step 1 - Origins
Our tale begins a long, long time ago, when only one of the three species existed, Hexagrammos octogrammus aka Hoc. Somewhere between 2.2 and 3.6 million years ago, a new line of fish diverged from the Hoc population. This was the common ancestor of Hag and Hot. For you biology geeks, this event was most likely an example of allopatric speciation, meaning that a primary driver of speciation was geographic isolation.
More time passed, and another split occurred, this one somewhere in the range of 1.2 to 2.0 million years ago, giving rise to Hag and Hot. Again for biology geeks—this event seems to be an example of sympatric speciation, meaning the Hag-Hot split occurred within the same geographical range. (3)
The resulting species distribution is depicted in Figure 1 above.
So Hoc came first. Then a common ancestor of Hag and Hot, with the final step being Hot breaking off to go off on its own.
There is more to the story than this executive summary, but that’s the basic understanding we need to move forward.
Step 2 - Hanky-Panky
Remember the Linnaean system of taxonomy that I bored you with above? It was to prepare you for this.
Recall that the lower down the family tree that two species find commonality, the closer they are in evolutionary/ genetic terms.
Hoc, Hag and Hot are all in the same genus, genetic cousins in a manner of speaking. This comes into play because at some point after the allopatric speciation process described above (geographical separation of Hag and Hot from Hoc), the species came into contact once again. Maybe this occurred once. Maybe it happened many times.
Whatever the case, this overlap provided the opportunity for hybridisation, i.e., hanky-panky between closely related species.
Hoc/ Hag hybrids appeared somewhere in the range of 100,000 to 1 million years ago. (4) The unusual thing about this is that the hybridisation was asymmetric. Hoc was and still is the maternal ancestor of all the Hoc/ Hag hybrids. (3) Genetic evidence showed that all Hoc/ Hag hybrids, which are reasonably common in the area of species overlap (the area labeled Hybrid zone in Figure 1), are related via only their mothers.
Also, all the Hoc/ Hag hybrids are female. No males.
It would be as if everyone in your extended family for the past 100,000 years or more shared genetic information inherited only from your mothers, and every member of your family seems to have been female, including you. Head scratcher, no?
In addition to Hoc/ Hag hybrids, researchers also found crosses between Hoc and Hot. Here too, in all instances, the maternal ancestor of all the Hoc/ Hot hybrids was Hoc. Asymmetric once again. Asymmetric in exactly the same way. And also—you guessed it–all the Hoc/ Hot hybrids appear to be and always to have been female. Estimates for when the Hoc/ Hot hybrids first appeared fall in the range of 2,000 to 30,000 years ago, more recent than the Hoc/ Hag hybrids. (4)(5)
Finally, while Hag and Hot appear to have a thing for Hoc, crosses between Hag and Hot do not appear to have happened.
This is the point at which it would be natural to ask: “Wait a second. If there had been random liaisons among Hoc and Hag or Hoc and Hot individuals, shouldn’t there be both females AND males?”
One would certainly think so.
When you find something odd like this, it is often a hint, a clue to a bigger revelation.
Step 3 - Science
The answer to the question of “Why do all the hybrids have only maternal ancestors?” was addressed by a team of Japanese researchers not too long ago, with their results published in 2014.(2)
To get to the bottom of this conundrum, they collected fish in the wild representing all the permutations: Hoc, Hag, Hot, as well as Hoc/ Hag and Hoc/ Hot. They then did what must have been tedious and painstaking work—artificial breeding of fish.
They crossed each of Hoc, Hag and Hot with their own species (i.e., normal sexual reproduction); they crossed Hoc and Hag, and Hoc and Hot (i.e., to simulate the hybrid combinations found in nature); they backcrossed naturally occurring hybrids with both their maternal and paternal species (Hoc/ Hag hybrid x Hoc, Hoc/ Hag hybrid x Hag, Hoc/ Hot hybrid x Hoc, Hoc/ Hot hybrid x Hot, where the first abbreviation in each hybrid pair denotes the maternal species).
I know. That jumble of letters comes across like a gigantic Scrabble disaster. Don’t worry too much about it. Just understand that the researchers performed genetic mix-and-match with all possible permutations to see what happens.
I’ll refer you to the paper by Kimura-Kawaguchi et al. (2) if you want to study the results and look at the figures and charts (I can predict the exact number of you who will want to do this), but the punchline is this:
“These findings indicate the strong possibility that the natural hybrids reproduce via hybridogenesis. The hybrids of Hexagrammos are the first hybridogenetic system identified from marine fishes.”
Earth-shattering.
Step 4 - Boy Toys
Here is why that statement blew my mind.
Remember I mentioned asexual reproduction earlier? I did so because asexual reproduction might explain female-only lineages. Something like parthogenesis or gynogenesis could have resulted in females producing only females producing only females, and so forth.
But recall that asexual reproduction, while having the advantage of efficiency (not needing or almost not needing males), has the inherent risk of vulnerability due to lack of genetic variation. While not impossible for an organism as complex as a fish, asexual reproduction as the primary/ only means of reproduction would not necessarily be the optimal or most evolutionarily stable situation.
What the scientists worked out in the 2014 paper and additional research since is that the Hoc/ Hag and Hoc/ Hot hybrids employ a trick known as hemiclonal reproduction, or hybridogenesis.
Hoc/ Hag females spawn with Hag males to produce a clutch of fertilised eggs. The resulting fish are all Hoc/ Hag female. Always. When those females grow up, they prepare eggs and find a Hag male to spawn with, but—here is the key—their eggs contain only Hoc DNA, i.e., genetic material from only the maternal side, not from both parents as would be the case with normal sexual reproduction.
The adult females' cells contain Hag DNA from their fathers, but the females discard that genetic information in the process of making eggs. So the females’ Hoc DNA is passed to their eggs to combine with male Hag DNA, but in each subsequent iteration, Hag DNA is sorted out again and dumped when it comes time to make eggs.
To put this another way, Hoc/ Hag females can be viewed as sexual parasites. They use Hag males for procreation. They use the males’ DNA to make healthy, viable babies. But they discard this DNA when they make eggs.
The result is that the Hoc genome within the Hoc/ Hag female perpetuates itself from generation to generation through a line of all females that use Hag males for (a) disposable DNA, and (b) protection of eggs.
In return for their diligence, fidelity and hard work, the males get nothing.
Ouch.
Moreover, females are polyandrous (yup, fancy word to impress you). They spawn several times during the reproductive season, so they use and discard multiple males.
Double ouch.
Hoc/ Hot hybrid females behave in exactly the same manner, except that they make use of Hot males instead of Hag.
Munehara et al (5) report that the Hoc/ Hot hybrid line was most likely the result of host-switching by a Hoc/ Hag fish, not of a direct hook-up between Hoc and Hot. In other words, a tryst between Hoc and Hag created the Hoc/ Hag line of super-females. Later, one of those females switched her preferred choice of boy toys from Hag to Hot. This fits with the researchers’ belief that Hoc/ Hot hybrids first appeared considerably later than the Hoc/ Hag female line.
Why this change of hosts happened is up for debate. Hot males are larger than Hag males and can be relatively more aggressive in defending their nests and territories. So perhaps that had something to do with it. But then again, maybe it was just random.
To the best of the researchers’ knowledge at this point, that host-switch from Hag to Hot happened only once. It is certainly possible that the switch took place again, but there has been no evidence found yet to support that view.
Step 5 - Renewal
The final part of this story is the maternal backcross—when a Hoc/ Hag hybrid female spawns with a male Hoc instead of male Hag. Or in the case of Hoc/Hot, with a Hoc male instead of Hot.
This happens from time-to-time. It serves a crucial purpose.
To understand why this is useful for the hybrids, we have to go back once again to the discussion of asexual reproduction. As we’ve learned so far, the all-female hybrids pass down only their maternal Hoc DNA to each generation of eggs. To reduce risk of vulnerability due to genetic uniformity, they use Hag or Hot DNA from males for each generation (which, remember, is discarded for the next generation).
This using of Hag or Hot males helps to mitigate the primary drawback of asexual reproduction, but it still leaves the maternal Hoc DNA constant over time (ignoring natural mutation).
The ingenious solution to this potential genetic bottleneck is maternal backcrossing. When a Hoc/ Hag hybrid spawns with a Hoc male (instead of Hag), gametes mix genetic material as they would with normal sexual reproduction, and—here is the crucial point—the recombined genes get passed on when the resulting babies grow up and spawn.
In other words, DNA from the female and the male combine in the egg as normal, resulting in babies that look like normal Hoc fish (Hoc/ Hag fish look a little bit different). More importantly, some of those babies will possess the genetic X-factor that can produce the female Hoc/ Hag hybrids, so they are carriers. Should one of those carriers be female and spawn with a Hag male, a new line of female Hoc/ Hag hybrid results, with genetic variation introduced via the maternal backcross Hoc male. Ditto that process for Hoc/ Hot.
It is like supercharging and renewing the wonder women genome from time to time.
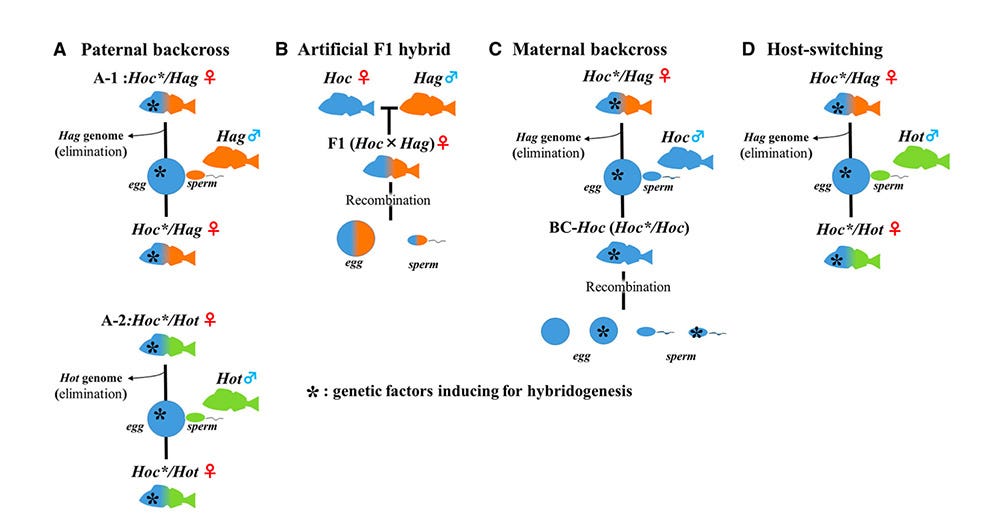
That’s a lot to wrap your mind around. It’s confusing. It took me many days of poring through papers, scratching my head, feeling stupid(er than normal).
This diagram might help. It spells out the permutations related to understanding hemiclonal reproduction. The asterisk symbol represents the genetic X-factor for creating the all-female wonder women hybrids.
A. Paternal backcross is the basic scenario of how hybrid females use Hag or Hot males.
B. Artificial F1 hybrid shows the result of lab-created hybrids, i.e., normal hybrid fish with no hemiclonal capability (thus demonstrating the existence of a genetic X-factor in the naturally occurring Hoc/ Hag and Hoc/ Hot super females).
C. Maternal backcross is what I just described above, the process of renewing the all-female hybrid genome.
D. Host-switching shows what likely happened the one time when a Hoc/ Hag female decided she preferred Hot males.
One last thing to note—there are fewer than 20 animal genera for which hemicolonal reproduction has been documented.(6) Hexagrammos hybrid females are the only example among marine species. Cool, eh?
Kevin has just rushed out to defend the perimeter. He chases fish after fish, most of which turn tail in full-blown panic, no doubt wondering, “What the heck just happened?”
Kevin, you see, takes his job seriously. He charges out at the slightest hint of trouble, even if the suspected territorial incursion is far beyond the distance at which a fish could reasonably be considered a threat.
Kevin is a good father.
Which might explain why his eggs were always in pristine condition; perhaps even why there were no hemiclonal eggs?
Kevin became Kevin because after that first encounter this season, I realised that he was special. He was outgoing. Inquisitive. Interactive. Alert. Friendly (to humans). He was yellow, a colour that contrasts perfectly against the blue of the ocean. He made me laugh. A lot. In other words, he was obviously a minion.
So I named him Kevin. I also cross-checked with photos from 2019 and realised that I had spent a few hours with Kevin last year (first two photos above). That might be why he felt familiar.
There were also two other nesting males nearby, who naturally became Stuart and Bob.
Together, this trio of aquatic minions kept me occupied for weeks. Their personality differences were fascinating.
Kevin you know about. I spent the most time with him.
Stuart was somewhat wary of people, but he was reasonably attentive to his eggs. He’d stick around and chase off fish, but not all the time, and he wasn’t as comfortable with human presence as Kevin. Sato-san, who has spent many years observing these fish, thinks Stuart may have been caught on a hook by a recreational fisherman. There was evidence of a scar. The trauma may have shaken his confidence.
Then there was Bob, who was a bit of a yellow-belly (groan). Bob ducked for cover whenever anyone or anything approached.
What led me to spend hours trying to understand hemiclonal reproduction (and by the transitive property, has caused you to devote a good chunk of your life to this topic if you’ve read this far) is noticing these eggs:
Hexagrammos otakii eggs come in many colours, but not this combination of purple and orange, which is reminiscent of Hexagrammos agrammus eggs. These eggs were among those in Bob’s nesting site. They are about 3mm (0.12 inches, aka super-duper-teeny-tiny) in size, and they immediately stood out.
There were similar eggs among Stuart’s collection. See the purple ones in the middle?
I mentioned these eggs to Sato-san. He told me about Munehara-san and his colleagues, people who’ve devoted much of their lives to solving the mysteries of the female-only hybrid species. They were based next door, so I asked for relevant research papers.
The purple eggs in the two photos above were spawned by Hoc/ Hot females. Stuart and Bob were assigned the job of watching over them until they hatch. All the babies from the purple eggs will be female. All will discard Stuart and Bob’s DNA when they produce eggs.
Bob was so bad at this job that the majority of his eggs seem to have been pilfered by hungry mouths. Stuart was better, but there were the occasional patches of missing eggs, holes formed in the size and shape of a fish’s mouth.
There were no such hybrid eggs in Kevin’s collection. There were also never any eggs missing from his nest.
Is it possible that Kevin had the ability to tell the difference between an actual Hot female and a sneaky Hoc/ Hot who was just out to use him?
Some weeks after I left, I received news that Kevin’s eggs had all successfully hatched. His work done for the season, Kevin returned to deeper waters, where he will stay, along with others of his species, until next year.
I do not know Kevin’s age, nor do I know how long these fish live. With a bit of luck, perhaps we will meet again.
I am certain that he and the wonder women of Hexagrammos have many more secrets left to share.
Wishing you a wonderful holiday season.
ars gratia scientiae
References:
(1) What A Fish Knows, The Inner Lives of Our Underwater Cousins, John Balcomb. Part VI: How a Fish Breeds, Parenting Styles.
(2) Kimura-Kawaguchi MR, Horita M, Abe S, Arai K, Kawata M, Munehara H. Identification of hemiclonal reproduction in three species of Hexagrammos marine reef fishes. Journal of Fish Biology (2014) 85, 189–209. https://doi.org/10.1111/jfb.12414
(3) Crow, Karen, Munehara, Hiroyuki, Bernardi, Giacomo. Sympatric speciation in a genus of marine reef fishes. Molecular Ecology (2010) 19, 2089–2105. https://doi.org/10.1111/j.1365-294X.2010.04611.x
(4) Suzuki S, Miyake S, Arai K, Munehara H. Unisexual hybrids break through an evolutionary dead end by two-way backcrossing. Evolution 2020 Feb, 74(2):392-403. https://doi.org/10.1111/evo.13903
(5) Munehara H, Hirota M, Kimura-Kawaguchi MR, Yamazaki A. Origins of two hemiclonal hybrids among three Hexagrammos species (Teleostei: Hexagrammidae): genetic diversification through host switching. Ecology and Evolution 2016; 6: 7126–7140. https://doi.org/10.1002/ece3.2446
(6) Lavanchy G, Schwander T. Hybridogenesis. Current Biology 29, R1–R15, January 7, 2019. https://www.cell.com/current-biology/pdf/S0960-9822(18)31545-8.pdf